mHealth Suite Platform – DiGA Development, DTx & Pharma Companion Apps
Our Platform Approach
Your CE-certified mHealth product on the market in just 3 months
At DUX Healthcare, we are redefining how regulated digital health products are built. As a platform provider, we deliver more than code – we provide the regulatory foundation, modular software architecture, and long-term operational reliability.
Our innovative manufacturing framework for Software as a Medical Device (SaMD) enables pharmaceutical companies, DiGA and DiPA manufacturers, startups, and health tech innovators to launch faster, scale smarter, and maintain seamless compliance.

Our Claim? Delivered!
From startups and DiGA manufacturers to pharmaceutical companies
The mHealth Suite
The technology solution that adapts to your specific medical purpose
Scalable Platform for Regulated Digital Medical Devices (SaMD)

Unmatched Speed

Seamless Scalability

Automated Documentation
Revolutionary Product Development –
Three approaches tailored to your starting point.

Pharma Companion Apps

DiGA Development & DiGA Transfer

DiPA Development
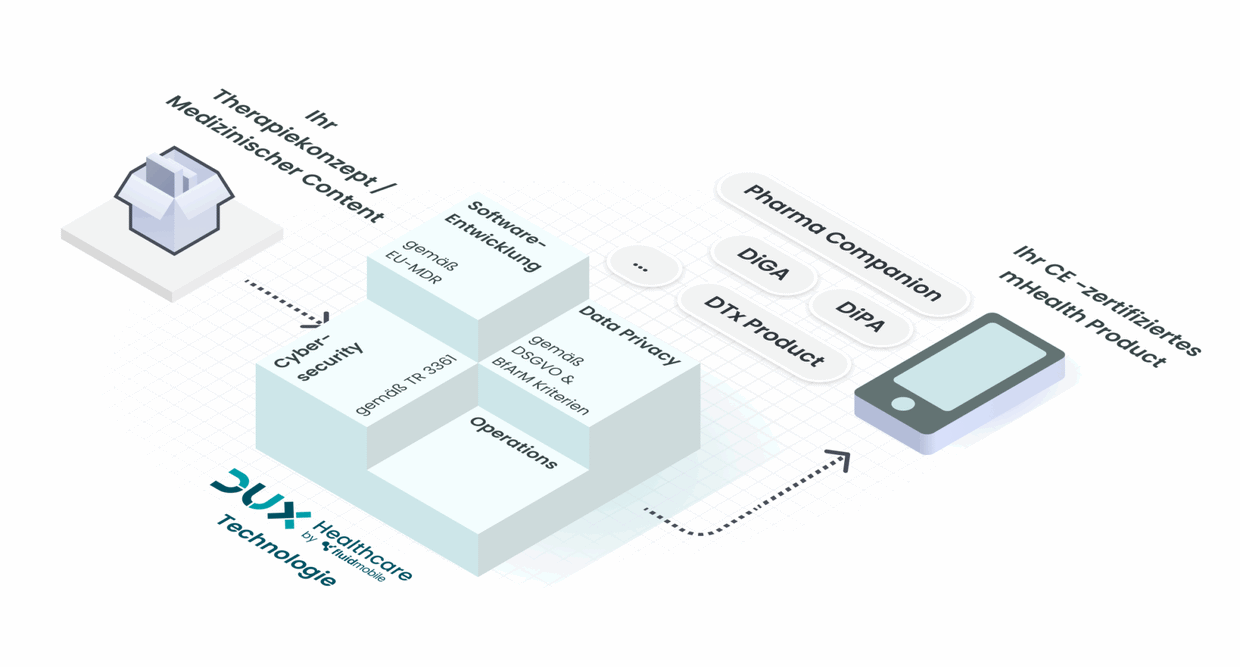
You provide the therapeutic concept – we deliver the market-ready digital product.
Our platform combines indication-agnostic software with individual customization.
The process starts with the therapeutic approach: our partners or clients provide the medical concept and domain expertise.
In parallel with content creation, technical implementation begins. Thanks to our highly modular platform architecture, product development starts immediately.
After approximately eight weeks, we deliver a complete MDR-compliant documentation package, enabling clinical evaluation and CE certification to proceed.
Launch can take place as early as three months after development begins.
On request, DUX Healthcare and its partners can support certification and market entry – alternatively, the client can act as the legal manufacturer themselves and bring the product to approval using the documentation provided.
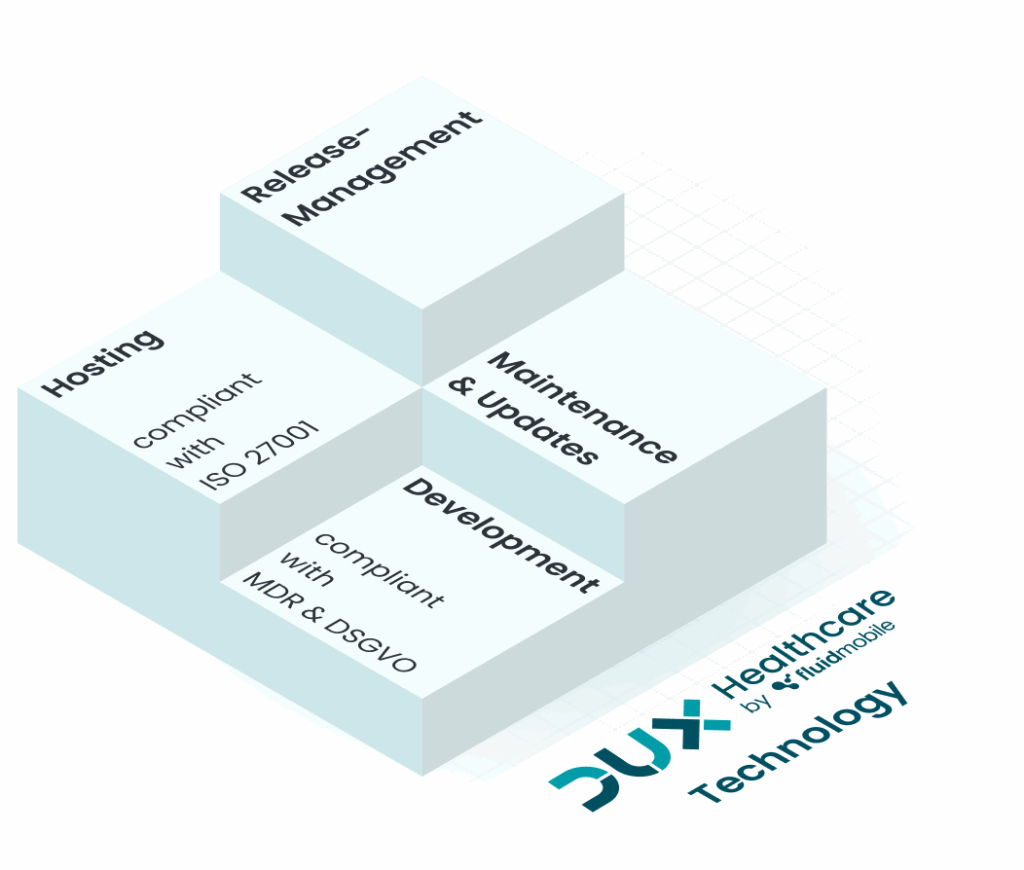
About DUX Healthcare
What sets us apart

ISO 13485 Certified

ISO 27001-Compliant Hosting

14 Years of Expertise

From fluidmobile to DUX Healthcare – what's changing and what stays the same
Since early 2026, we operate as DUX Healthcare – formerly fluidmobile GmbH. DUX Healthcare GmbH (formerly fluidmobile GmbH) is your legal contracting partner, data processor, and compliance entity.
For you, only the name changes – our contracts and our focus on certified products remain the same.
Looking for a solution to your mHealth challenges?
Regulatory complexity, data security, or rapid scale-up – the path to a finished mHealth product can be long and challenging.
With the mHealth Suite, we support pharmaceutical companies, health tech startups, and DiGA/DiPA manufacturers with product strategy, digital health, and marketing. We also evaluate the regulatory and strategic framework for your product.
Let’s turn your concept into a compliant, scalable product together.

