We Solve Challenges in DiGA Development
Challenge 1: Complex & Evolving Regulations
Challenge 2: High Upfront Costs, Uncertain ROI
Challenge 3: No Room for Trial & Error
Challenge 4: Lack of Scalable Infrastructure
Our Promise
We typically bring your application to MDR-compliant product development and completed CE certification in just three months after project start — as a Class I or IIa medical device.
Our platform meets all technical requirements of the DiGAV, the BfArM data protection review criteria, and BSI TR-03161, and is ready for ePA connectivity and Health ID integration.
This gives you the technical and regulatory foundation for your DiGA — whether as a new development or a rapid transfer of existing solutions in just 2–3 months. We can also support the evaluation of the positive care effect with clinical study partners upon request.
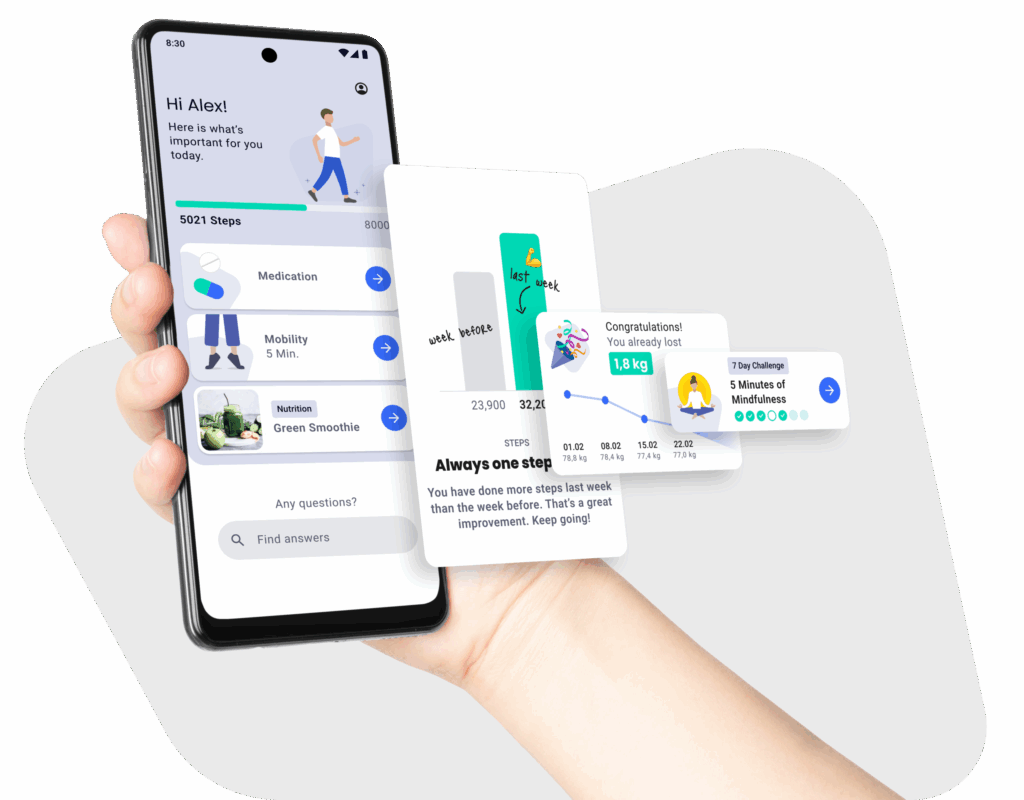
How DiGA Manufacturers Benefit from DiGA Development with the mHealth Suite

DiGA Transfer

MDR & Documentation Built In

Stay Listed & Stay Secure
Want to Learn More?
DiGA Development with the mHealth Suite
- Innovative manufacturing framework for Software as a Medical Device (SaMD)
- Scalable development platform for CE-certified medical devices — for DiGAs, Pharma Companion Apps, and Digital Therapeutics (DTx)
- Auto-generated MDR-compliant Technical Documentation audit-ready at the push of a button
- Automated Software Verification & Validation (V&V) for greater compliance and fewer risks
- 80+ pre-validated DTx modules individually recombined and tailored to your medical intended purpose
- Indication-agnostic flexibly adaptable to your target indication — from chronic diseases to preventive care offerings
- Subscription model transparent, predictable costs with full scalability — ideal for startups, pharma, DiGA, and DiPA manufacturers
- Scalability in focus from MVP to international rollout
FAQ on the mHealth Suite and DiGA Development
Is the mHealth Suite a platform I can use myself?
No — the mHealth Suite is not a self-service tool but our internal production framework that we use to develop your digital health application. Think of it as our “digital production line” for CE-certified medical apps, DiGAs, and Companion Apps — developed by fluidmobile GmbH for our own use.
You don’t receive a platform you have to operate yourself, but rather a finished product built on our proven infrastructure — including development, operations, and regulatory assurance. You provide the therapy concept — we take care of implementation, certification, and maintenance.
What does the mHealth Suite offer — and how does it support my product idea?
The mHealth Suite is a scalable platform used by DUX Healthcare to develop CE-certified digital health applications in just three months.
You benefit from over 80 pre-validated DTx modules, automated Technical Documentation, and integrated regulatory compliance (MDR, DiGAV, BSI TR-03161, BfArM data protection review criteria). The scalable development platform enables predictable costs through a transparent subscription model and drastically reduces development and certification time as well as maintenance effort.
Whether DiGA, DiPA, Pharma Companion App, DTx, or lifestyle product — the solution is indication-agnostic and can be customized to your therapy content. You provide the therapy concept — we deliver the regulatory infrastructure.
How quickly can I go live with my digital product?
A typical project timeline with a nearly complete concept is around 3 months from kick-off to completion of product development including Class I and IIa CE marking. A lifestyle MVP can typically be CE-ready within 8 weeks of project start.
Pre-validated components and automated documentation reduce development and certification time by up to 80% compared to conventional approaches. A transfer of an existing DiGA can be completed in 2–3 months, while a full DiGA development including clinical study and DiGA application with BfArM takes approximately 12 months.
How does the mHealth Suite pricing model work?
The mHealth Suite offers a transparent and predictable cost structure. After a one-time setup fee for configuration and integration, you pay a monthly subscription fee. Exact flat-rate pricing is available upon request.
The minimum term of the subscription model is 12 months. The monthly subscription fee includes hosting, maintenance, compliance services, support, and ongoing development. Additional services such as external CE certification or exclusive modules can be added optionally.
What types of health products can DUX Healthcare develop for me?
With the mHealth Suite, we develop digital health solutions of all kinds:
- Health software & lifestyle apps — even without CE, ideal for prevention, wellbeing, or initial MVPs with a later upgrade path
- Medical device software / medical apps — Class I, IIa, or IIb, fully MDR-compliant, CE-certified according to ISO 13485 and ISO 27001
- Pharma Companion Apps — digital companions for Rx products, OTC, or Orphan Drugs
- Digital Health Applications (DiGA) — in compliance with DiGAV, BSI TR-03161, and BfArM data protection review criteria
Who takes on the role of Legal Manufacturer?
Does the mHealth Suite really cover all regulatory requirements for DiGA and DiPA?
What does DUX Healthcare handle after my app launches?
Can I expand my product with new features or indications later?
Will my app be kept technically up to date?
Is it possible to scale the app for additional indications or target groups?
Can custom modules be added?
Contact our experts

