For Pharma Partners
Pharma Companion Apps for Rx, OTC & Orphan Drugs
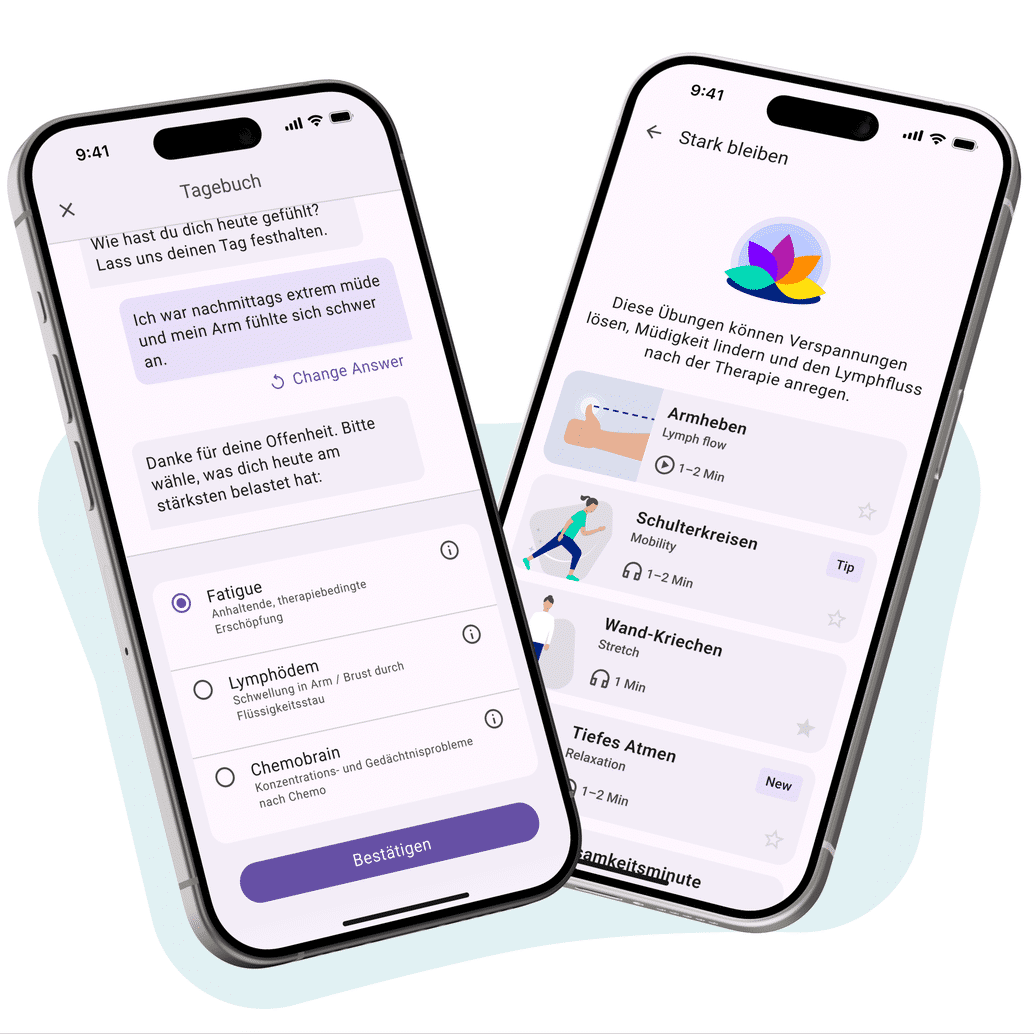
Companion Apps as a Bridge Between Therapy, Patients & Real-World Evidence
Creating proximity where others can't reach.
Rare diseases require targeted communication, complex care management, and high-quality patient support. This is exactly where digital Companion Apps make a difference: they guide patients individually through therapy pathways, facilitate education, and connect healthcare professionals with patients.
At the same time, they enable Real-World Data collection – GDPR- and MDR-compliant. Your solution for pre-marketing in compliance with HWG (German Pharmaceutical Advertising Act) – scalable, certified, ready to deploy.
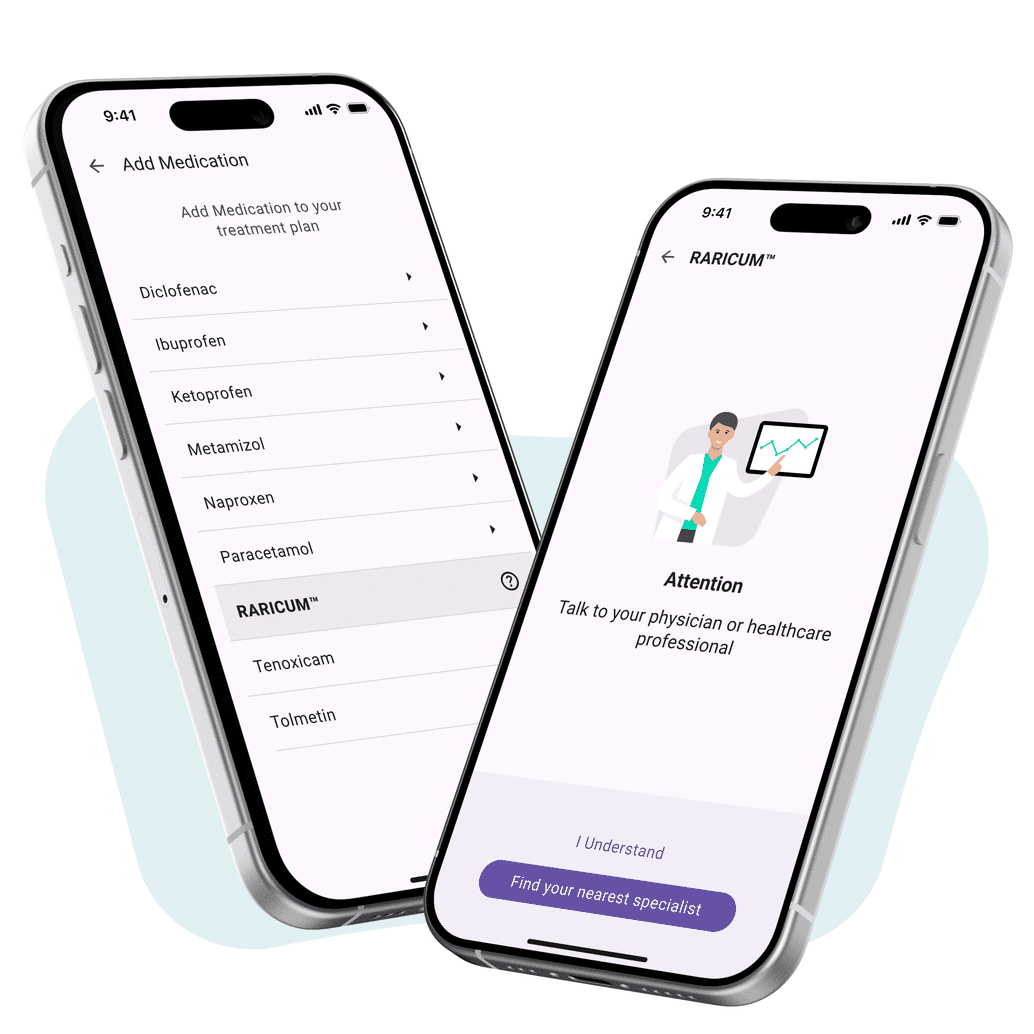
Your solution for pre-marketing in compliance with HWG – scalable, certified, ready to deploy.
Rx – Digital Market Launch with a Head Start
Digital companions that turn one-time purchases into real brand experiences.
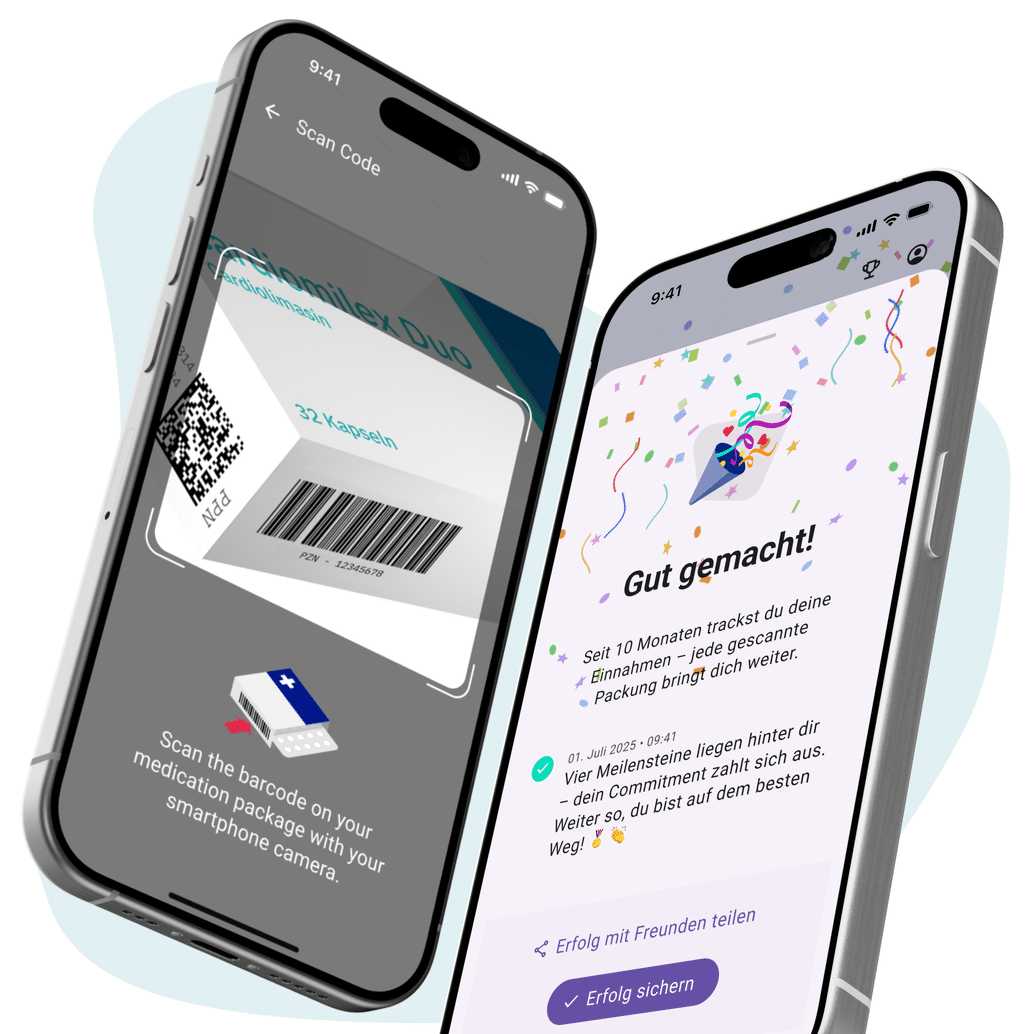
OTC – More Purchases. More Loyalty. More Brand.
We know the market's hurdles. And we have the right platform to overcome them.
Your Challenges?

Rx
- HWG (German Pharmaceutical Advertising Act) prevents early product communication
- MDR and approval obligations block dynamic execution
- Physicians and patients are often unprepared at launch

OTC
- Little brand loyalty – high interchangeability
- No dialogue after purchase – no loyalty
- Low user engagement and no continuous data utilization

Orphan Drugs
- Very small target groups – hard to reach
- Complex therapy pathways – high information needs
- Low awareness – minimal market attention
The Smart Pharma Companion App for Your Medication
Our Promise
Whether Rx, OTC, or Orphan Drug – we make your vision digital. With the mHealth Suite, you bring a Pharma Companion App to market that delivers more: Early patient activation, recurring brand engagement, Real-World insights.
CE-certified, HWG-compliant, MDR-ready – faster, more scalable, and more strategically valuable than ever before. For all phases. For all markets. For your growth objectives.
Maximize the Potential of Your Medication with Pharma Companion Apps
How Pharmaceutical Companies Benefit from the mHealth Suite

Fast Time-to-Market

Long-Term Regulatory Assurance

Pre-Marketing Innovation
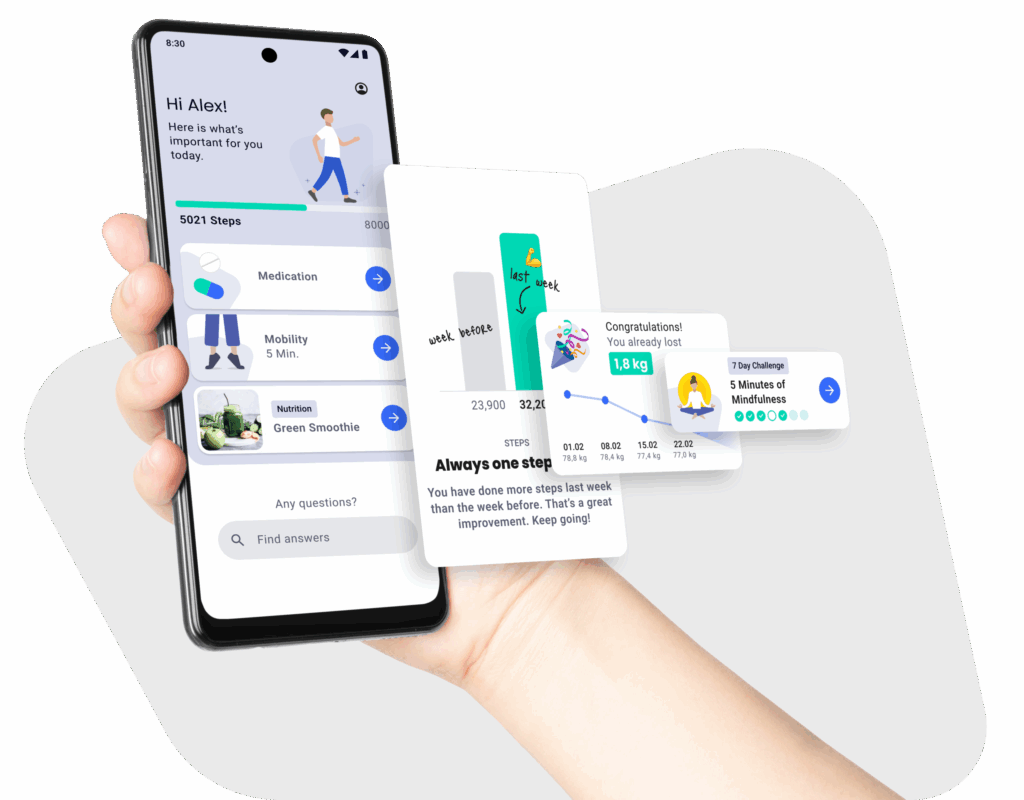
Certified, Secure, and Compliant App Development
The mHealth Suite
DUX Healthcare is ISO 13485 certified and has deep regulatory expertise – including MDR, BSI TR-03161, and GDPR compliance. Our platform meets the highest standards for security, data protection, and scalability.
Over 80 DTx modules deliver fast, cost-efficient, and highly customizable solutions. Instead of developing every feature from scratch, we combine existing modules tailored to each indication – quality and compliance standards are built in from the start.
CASE STUDY: My-formoline L112 - Companion
The My-formoline app is your reliable partner on the path to a healthier and more active lifestyle – specifically designed to support the use of formoline L112. As a medical device, the app offers numerous features that guide you step by step toward reaching and maintaining your weight loss goals.
Features include: Weight tracking, weekly overviews, step counter, hydration reminders, intake reminders, and a comprehensive knowledge library.
FAQ
Is the mHealth Suite a platform I can use myself?
What does the mHealth Suite offer?
How quickly can I go live?
How does the pricing model work?
Who assumes the role of Legal Manufacturer?
Does the mHealth Suite cover all regulatory requirements?
What happens after launch?
Can I add features later?
Is scaling possible?
Can custom modules be added?
Contact our experts

